  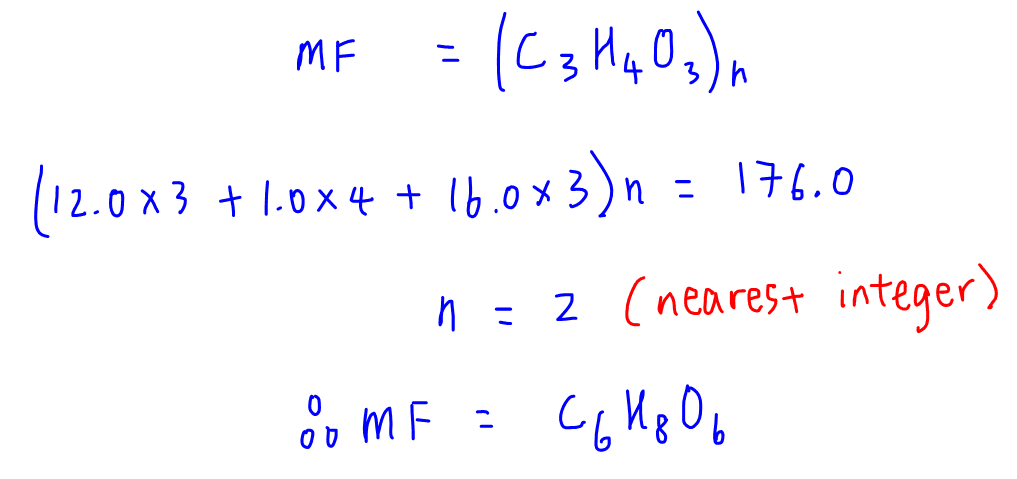
If you would like to learn more about the other chemical calculator that gives instant results, stay tuned to Onlinecalculator. 2.67 g O / 16 (g/mol) O 0.1669 mol O If we now divide the found numbers of moles by the minimum value of 0.1665 and round to whole values, then we get the following indices for the empirical formula: 1, 2, 1. Therefore, the empirical formula is Ca 3H 522O 5 
It takes the chemical composition percentage and gives the empirical formula as result easily. Molar mass of Ca = 40.078, O = 15.9994, H = 1.00794ĭivide all components by the smallest valueĭivide by fractional component of each mole value An empirical formula calculator is an online tool that allows finding the empirical formula corresponding to the chemical composition. Question: Calculate the empirical formula for composition Ca = 13.5%, H = 67.5%, O = 10.8%. these measurements permit the calculation of the compounds percent. The major difference is empirical formula gives the simplest ratio of atoms involved in the compound, the molecular formula gives the total number of atoms of an element in the compound. Given the chemical formula of the substance, we were able to determine the amount. The molecular formula of glucose is C H O 6, but its empirical formula is CH 2O. It represents the percentage of an element in the compound. The relative number of atoms of every element in the chemical compound is given by this empirical formula. That means the percents can be expresse as mass: Na. Enter an optional molar mass to find the molecular formula. Calculate empirical formula when given percent composition data 1) Assume 100 g of the compound is present. CoolGyan’S online empirical calculator tool makes the calculation faster, and it displays the formula in a fraction of seconds. To calculate the empirical formula, enter the composition (e.g. Empirical Calculator is a free online tool that displays the empirical formula for the given chemical composition. In chemistry, an empirical formula in chemical composition is the simplest positive integer ratio of the atoms present in that chemical compound. Elemental analysis is a useful qualitative analysis technique since it allows us to check if a sample is consistent with a given molecular formula. Empirical Formula Calculator - ChemicalAid 1 day ago Show details This program determines both empirical and molecular formulas. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
